The Spectrum of Diabetes
Are the two 'types' really separate conditions?
Disclaimer: nothing in this article is to be taken as medical advice. The information provided here is to be used purely for informational and educational purposes.
Diabetes mellitus, usually simply referred to as ‘diabetes’ refers to several conditions that are characterised by increased blood glucose concentration. Although they share the same name, ‘diabetes insipidus’ refers to a separate condition. In this article, we will be focusing on ‘diabetes mellitus’.
Diabetes mellitus is defined by the WHO as “a chronic disease that occurs either when the pancreas does not produce enough insulin or when the body cannot effectively use the insulin it produces”. Without insulin, the body cannot access the glucose it needs to function properly. This results in hyperglycemia (high blood sugar), which in turn can lead to, among other things, a life-threatening condition known as diabetic ketoacidosis, or ‘DKA’.
Insulin is a peptide (small protein) hormone produced by pancreatic beta (β) cells, located inside the pancreatic islets, or ‘islets of Langerhans’. Once released from these cells, it flows through the blood, and binds with insulin receptors located on cell surfaces. It is the ‘key’, that ‘unlocks’ the glucose channels (GLUT4 in this case), thus allowing glucose granules circulating in the blood to get into the cells, used in turn by the cells for the production of energy.
Beta cells are triggered into secreting insulin through glucose-stimulated insulin secretion, or ‘GSIS’. This process is described in more detail here, and in the embedded video below. In short; glucose granules enter beta cells through their dedicated glucose channel; GLUT2 in this case. Any glucose taken up by these cells is ‘phosphorylated’ (combined with a phosphate molecule), creating glucose-6-phosphate, used for the production of adenosine triphosphate, or ATP. ATP is a compound used by cells throughout the body to provide energy. In the beta cells, production of ATP closes the beta cell’s potassium channel, which in turn opens the calcium channel. Calcium flows in, and the expulsion of insulin from the cell is triggered.
The insulin produced by these cells then flows through the liver, which uses up more than 50% of it for the purpose of storing glucose as glycogen. The remaining insulin exits the liver via the hepatic vein, after which it is distributed to the rest of the body through arterial circulation.
Insulin binds to cells through insulin receptors located on the outer part of the cell membrane, that are made up of two molecules linked by disulphide bonds. These molecules include an ‘alpha-chain’ and a ‘beta-chain’; the alpha-chain is located entirely outside the cell, and contains 37 cysteine residues (sulphur-containing amino acids), most of which form ‘intra-chain’ disulphide bonds. The beta-chain contains 10 cysteine residues, and spans the entire cell membrane; part of it is inside the cell, and part of it, outside. Insulin binds with the alpha-chain portion of the receptor, bringing the two halves together. This results in the two sides of the beta-chain portion of the receptor located inside the cell, to make contact, and triggers a ‘signalling cascade’ (a series of chemical reactions), that eventually results in an instruction for the cell to allow the entry of glucose granules from the blood into the cell, through the cell’s GLUT channels.
This process was first described by a team of researchers in 2014, and the following animation explains what is going on in more detail.
The inability of the body to produce insulin to activate these receptors, or the receptors not working properly when binding with the insulin, is what is said to differentiate the two main forms of diabetes; ‘Type 1’ (T1) and ‘Type 2’ (T2).
Type 1
Also known as ‘insulin-dependant’, ‘juvenile’ or ‘childhood-onset’, this is said to be an ‘autoimmune condition’, whereby the body “mistakenly attacks and destroys insulin-producing beta cells in the pancreas”. Islet autoantibodies are created by B cells (another type of cell), which in turn bind to the surface of beta cells, marking them for destruction by T cells. The presence of these antibodies is said to be one of the hallmark features of Type 1.
Destruction of beta cells means the body cannot create its own insulin. ‘Exogenous insulin’ (insulin that is injected) thus needs to be provided by means of subcutaneous injections, multiple times a day. It is a debilitating condition that requires the continuous monitoring of blood glucose levels, which are primarily affected by eating. Insulin needs to be dosed carefully; too much can result in ‘hypoglycemia’ (low blood sugar), whilst too little can result in ‘hyperglycemia’ (high blood sugar).
As previously mentioned, any insulin secreted by the pancreatic beta cells flows through the liver. Insulin that is injected bypasses this system, resulting in less glycogen being stored; “the deficiency of insulin seen in type 1 diabetes leads to a decrease in liver and muscle glycogen levels”. This makes blood sugar levels harder to manage. If energy reserves are not available in the form of glycogen, major organs begin to be affected, such as the brain. In emergency cases, low blood sugar may require a glucagon injection. Glucagon is produced by the pancreatic alpha cells, and helps bring blood sugar levels back up to where they should be, by either preventing the liver from taking glucose out of the blood and turning it into glycogen, or triggering the liver to release glucose from its glycogen reserves.
According to the WHO; “in 2017 there were 9 million people with type 1 diabetes; the majority of them live in high-income countries. Neither its cause nor the means to prevent it are known”. According to the CDC, certain individuals have genes that make them more likely to develop the condition, however these alone are not sufficient; “a trigger in the environment, such as a virus, may also play a part in developing type 1 diabetes. Diet and lifestyle habits don’t cause type 1 diabetes”.
Research published in 2017 found that children exposed to ‘enteroviruses’, including ‘poliovirus’ and ‘coxsackievirus’ (hand, foot, and mouth) were more likely to develop Type 1 diabetes. ‘Sars-COV-2’ also appears to be one such trigger. In the spring of 2020, physicians in New York City (the U.S. epicenter of the ‘COVID pandemic’ at the time), noticed a considerable number of people hospitalised with ‘COVID’ that had hyperglycemia. Endocrinologist Paolo Fiorina from Harvard Medical School reported that of 551 patients hospitalised with ‘COVID’ in Italy, nearly half became hyperglycaemic. Peter Jackson, a biochemist at the Stanford University School of Medicine, estimated that up to 30% of patients with severe ‘COVID’ may develop diabetes. In London, a study found that hospitals across north-west London saw an 80% increase in cases in children during the peak of the ‘pandemic’. A literature review published in November 2020 found that 14.4% of hospitalised ‘COVID’ patients went on to develop diabetes.
Type 1 is also said to be caused by other conditions, such as haemochromatosis, which according to the NHS, is “an inherited condition where iron levels in the body slowly build up over many years”. This is referred to as ‘secondary diabetes’.
According to the CDC; “[Type 1] usually develops in children, teens, and young adults, but it can happen at any age”. Indeed, a case report published in 2022 details the case of a ‘Newly diagnosed type 1 diabetes in a 93 year old’. It is interesting to note that the individual in question “had a background of primary hypothyroidism and vitamin B12 deficiency”. Her Type 1 diagnosis was confirmed by the presence of autoantibodies. Interestingly however, this doesn’t appear to be a reliable marker given that “a number of patients with type 2 diabetes are GAD antibody positive”.
A literature review published in 2021 notes that “more than half of all new cases of type 1 diabetes occur in adults”. The study however notes that “the proportion would likely be higher if autoimmune cases not requiring insulin initially were classified as type 1 diabetes”. This type of diabetes is referred to as ‘adult-onset autoimmune diabetes (AID), which is subdivided into two possible types: “classic type 1 diabetes mellitus with insulin requirement just after diagnosis, and latent autoimmune diabetes in adults (LADA)”. According to Diabetes.org.uk, LADA “is a type of diabetes which seems to straddle type 1 and type 2 diabetes. Bits of it are more like type 1, and other bits are more like type 2. That's why some people call it type 1.5 diabetes or type 1 ½ diabetes”.
Type 1 diabetes it said to be irreversible and incurable, although some success has apparently been achieved using stem cell therapy. There are also some stories of individuals having allegedly recovered, such as that of Daniel Darkes. Experts are perplexed by his story which apparently involved the presence of a ‘rare gene’ that somehow facilitated his cure. Bart Roep, a clinical immunologist at City of Hope's Diabetes and Metabolism Research Institute in California, believes it is likely that Darkes was ‘misdiagnosed’, and had yet another type of diabetes known as ‘Maturity onset diabetes of the young’, or ‘MODY’. According to Roep, 5 – 11% of Type 1 diabetics could be misdiagnosed.
A case report published in 2019 in the British Medical Journal details the case of Sophie Fleming (35), who was misdiagnosed as having Type 1 when she was 8, and subsequently treated with insulin for 27 years. A C-peptide test put an end to her ordeal; she was able to stop taking insulin, and her condition can now be managed by taking sulfonylurea tablets. Although still not ideal, it is a vast improvement, as she says so herself. Sulfonylurea tablets are generally taken once or twice a day, with, or shortly before a meal. They are also given to Type 2 diabetics, sometimes alongside metformin in order to ‘stimulate the secretion of more insulin’.
C-peptide is produced alongside insulin in equal amounts, and is not a product of exogenous insulin. Therefore, testing for C-peptide levels is said to be a reliable way of measuring insulin secretion, even when the individual in question is taking insulin exogenously. Sophie’s new diagnosis provided a catalyst to offer “C peptide testing to all patients with type 1 diabetes in the Lothian region”. Given the potential for misdiagnosis, and the tremendous impact this can have on an individuals life, it is unclear why this sort of testing does not appear to be routinely done at the time of diagnosis.
Bart Roep also notes that “you only need 10 percent of your beta cells to supply sufficient insulin”. This is interesting, given that a literature review published in 2021 notes that “residual beta cell mass estimates in T1D, range from ~2% to 40%”, which would in theory mean that certain Type 1 diabetics should be able to produce their own insulin. We were unable to locate the source of the 10% figure provided by Roep, and experimental evidence appears to suggest this figure varies from case to case, and species to species. A paper published in 2008 concluded that “the extent of beta cell death was found to vary with age, whereby a 40% reduction at 20 years of age is sufficient to precipitate clinical symptoms of Type 1 diabetes mellitus. Thus, the statement that an 80–95% reduction in beta cell mass is necessary for the onset of hyperglycemia appeared to be an overestimate for most patients”.
The depletion of beta cell mass is said to be another hallmark of Type 1, and it had been previously thought that these cells were eventually completed destroyed. However, a literature review published in 2019 notes that “recent research has challenged the dogma that all beta cells are eventually destroyed in type 1 diabetes”. The various studies that make up the review showed that “persistent low-level beta cell function is relatively common in long-standing type 1 diabetes, particularly in people diagnosed as adults. A common feature of these studies is the small number of detectable beta cells and very low levels of endogenous insulin secretion. So, whilst the total number of beta cells in long-duration disease is vastly reduced, some persist and maintain the ability to produce and secrete insulin”. It is also interesting to note that beta cell mass appears to be depleted in both Type 1 and Type 2 diabetes; “autopsy studies report deficits in β-cell mass ranging from 0 to 65% in type 2 diabetes (T2DM), and ~70–100% in type 1 diabetes (T1DM)”.
Roep also mentions that there have been a couple of rare cases where a patient “had typical type 1 diabetes but could go through long periods without insulin injections”. This links to a case report published in 2012 in the BMJ that details the following case; “a child with classical newly diagnosed T1DM started on a gluten-free diet, remains without the need for exogenous insulin after 20 months”. The child was not diagnosed as having ‘celiac disease’, but according to the clinicians, “up to 10% of patients with T1DM also have celiac disorder and, interestingly, the two diseases are by far the most common observed combined, if diabetes is the first to appear, and seldom if celiac disease develops first and gluten-free diet is implemented”. Given that Type 1 is said to be incurable, the approach taken by the clinicians is curious. And why attempt a gluten-free diet? According to their report; “in gluten-free-diet-treated non-obese diabetic (NOD) mice never exposed to gliadin [a sulphur-rich component of gluten], the decline in incidence of T1DM was decreased from 61% to only 6%. The result has been confirmed by several groups and by corresponding studies in BB rats”.
Type 2
Type 2, also referred to as ‘non-insulin-dependent’, or ‘adult-onset’, is commonly thought to be a ‘metabolic disorder’ brought on by excess weight, resulting from diet and lack of activity; in short a ‘modern disease’ brought about by our ‘modern lifestyle’. According to the WHO, 95% of diabetics fall in this category.
According to a study published in 2011; “type 2 diabetes is in the process of being redefined as an autoimmune disease”, and as previously mentioned, the same autoantibodies as those that are said to mediate beta cell destruction in cases of Type 1, are sometimes detected in cases of Type 2. Because Type 1 is said to be a condition that occurs mainly in young people, and Type 2 in adults, it can happen that young people are mistakenly diagnosed as Type 1, as seen in the example of an overweight 15 year old Nigerian girl, initially diagnosed as Type 1 but later as Type 2. She was misdiagnosed due to her age; “the reason many physicians still misdiagnose T2DM in adolescents as type 1 diabetes mellitus for reason of age of onset”.
Excess weight doesn’t however, appear to always be a prerequisite for Type 2 to develop. A study published in Japan in 2012 found that “of 218 children who were diagnosed as having T2DM by a school urine glucose screening program in Tokyo, 24 were identified as being non-obese. None of the children had any evidence of β-cell autoimmunity or genetic disorders”.
Furthermore, a study published in 2017 found that misdiagnosis is a real problem; “more than 50% of patients diagnosed with type 1 diabetes after age 35 years were shown to have type 2 diabetes in long-term follow-up … Conversely, many older patients initially believed to have type 2 diabetes because of their age at diagnosis deteriorate rapidly and are subsequently found to have type 1 diabetes”. Former British Prime Minister Theresa May, is one such example. Dr Emily Burns of Diabetes UK is reported as having said: “we’d ask healthcare professionals to have this insight in mind: don’t rule out type 1 diabetes after the age of 30”. This no doubt explains why terms like ‘juvenile-onset’ appear to have by and large, been abandoned.
Because Type 1 and Type 2 are said to be completely different ‘diseases’, it is said that it is impossible for a Type 2 diabetic to become a Type 1 and vice-versa. However, “a person who originally receives a diagnosis of type 2 diabetes may still get a separate diagnosis of type 1 at a later date”. In other words, they can ‘change’ into each other through the ‘misdiagnosis’ exit clause.
Diabetic ketoacidosis is said to be another hallmark of Type 1 diabetes, however according to a literature review published in 2014; “its presence has been increasingly recognised in patients with type 2 diabetes and a newer entity called ketosis prone diabetes is also commonly recognised”.
Statistics, and a bit of history
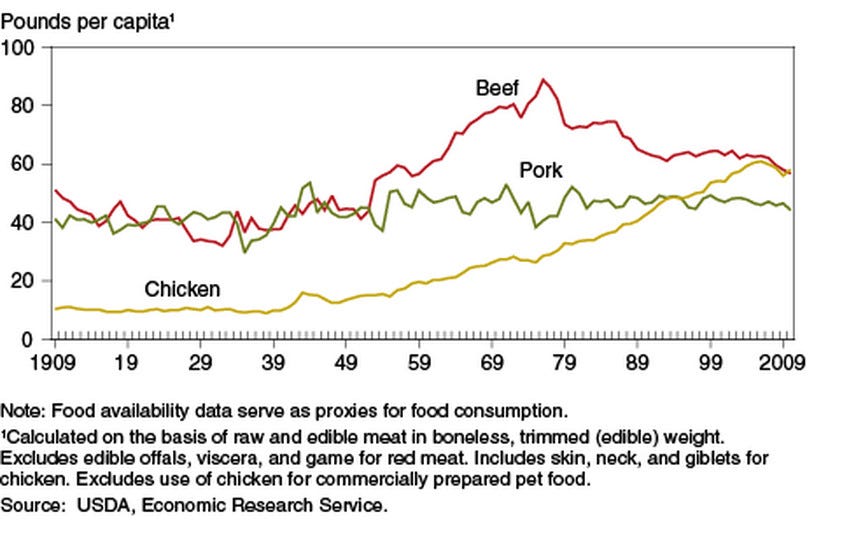
The incidence of diabetes is rapidly rising. In the US, the CDC’s 2020 National Diabetes Statistics Report estimated an almost 30% increase in Type 1 cases within the United States since 2017. According to the WHO, there are 422 million people worldwide with diabetes, and “the number of cases and the prevalence of diabetes have been steadily increasing over the past few decades”. In the UK, the number of people diagnosed has risen from 1.4m to 3.9m since 1996, although the true figure is thought to be closer to 4.8m. In November 2022, Diabetes UK reported that people under 40 diagnosed with Type 2 had jumped 23% from ~ 120,000 in 2016-17, to 148,000 in 2020-21.
Finding detailed, up to date statistics of the incidence rate of the different types of diabetes in different countries, and for all ages, has proven to be challenging. The top result in Google is for a league table published by Diabetes.org.uk in 2013, but that only provides the incidence rate in the age group 0 – 14. Finland, Sweden, Saudi Arabia, Norway, the UK, USA, Australia, Kuwait, Denmark and Canada make up the top 10 countries. A study published in 2022 provided some more up to date figures. This time, the incidence rate for Type 1 diabetes is provided for the age group 0 - 19, and it is the US, India, Brazil, China, Russia, Algeria, Germany, Morocco and Saudi Arabia that make up the top 10 countries. For Type 2, statistics are provided for the age group 20 - 79, and it is Pakistan, French Polynesia, Kuwait and New Caledonia, Northern Mariana Islands, Nauru, The Marshall Islands, Mauritius, Kiribati and Egypt that make the top 10. It is unclear why the prevalence rate for the two types is calculated for a specific age group, and not for the population as a whole, given that both types are said to occur in all age groups.
Diabetes Rising, a book written and published in 2010 by Dan Hurley, chronicles the rise of diabetes over the years. According to Hurley, “once upon a time, diabetes was rare”. Hippocrates apparently made no mention of it, but Hurley notes that the Ebers papyrus details a number of remedies for “correcting urine that is in excess”. Although “frequent and copious urination … has always been considered the hallmark of the untreated or under-treated disease”, Hurley also notes that “plenty of other disorders can cause [excess urination]”. The next ‘great treatise’ on diabetes was written in 1798 by physician John Rollo, by which point “it had become so rare that after seeing a single case”, Rollo “wrote that he did not see another for 22 years, even though he had traveled widely in the intervening years”. This led him to write to other physicians, asking them to share their reports, which he compiled in a book ‘Cases of the Diabetes Mellitus’. This according to Hurley, “was the first publication in history to prescribe a low carbohydrate diet for the disease”.
Hurley notes that “it is striking” that none of the descriptions provided by Rollo and his colleagues “emphasised the chief predisposing risk factor that we all now associate with type 2 diabetes: excess weight”. According to Hurley, this could be because Rollo and “his predecessors saw only type 1 diabetes”, although he then remarks that this seems unlikely, given that “all the early descriptions of diabetes are plainly of type 2, involving adults, not children”. He then goes on to reference the example of a child provided by Dr. Rollo, “which almost surely, would have been type 1”. The case in question was recounted in 1810 by New York surgeon Dr. Valentine Mott, in a report titled ‘An account of an extraordinary case of diabetes mellitus’. It involved a 9 year old boy, who died shortly after Dr. Mott first saw him – and this despite “Dr Mott’s best efforts”, which Hurley recounts without a trace of irony, “included bleeding the boy regularly, and prescribing him opium and arsenic”. The report itself is very interesting, and worth reading in full. Of note is the fact Dr. Mott noticed that the boy’s gums were “spongy and foul”, which sound like the symptoms associated with ‘scurvy’, which it would appear is caused by poisoning, as documented here. The ‘nutritional deficiency’ associated with the condition, is a symptom – not the cause.
It is also interesting to note that over time, paralysis ensued; “the numbness before mentioned, which began in the fingers of the left hand, gradually extended up the arm, as was observed whenever the convulsions were off, and by the time the left him, the whole arm was entirely useless, the mouth drawn to the opposite side, and the leg of the same side paralytic; in fact there was complete hemiplegia formed”. The boy eventually passed, “he was seized with great oppression in breathing, from an effusion of mucus into the bronchiae”. As we have previously discussed, ‘scurvy’ and ‘polio’ can sometimes be mistaken for each other, as perhaps best exemplified by a case report published in 1993 of a young boy who was initially diagnosed with ‘polio’, but later ‘scurvy’. As mentioned earlier, it would also appear there is an association between ‘polio’ and a subsequent diabetes diagnosis.
Another possibility, according to Hurley, is that “because untreated diabetes causes weight loss … physicians where seeing patients whose weight by the time they sought medical assistance, appeared anything but excessive”. However, he then notes that “weight loss has long been seen as a symptom in all kinds of diseases, including cancer and tuberculosis”. Hurley then offers us the following explanation; “we are faced with the mystery then, that pre-industrialist physicians like Dr. Rollo and Dr. Willis, never noticed that the leading risk for factor disease is obesity. Rather than suspecting them of being dull-witted when it comes to something so obvious, we need to confront the more likely, and to our modern sensibility quite surprising, fact that most diabetics in those days did not weigh more than the average person”. Thus, Hurley concludes, “whilst diabetes is certainly exacerbated by weight, it is ultimately not caused by it”.
Hurley also mentions ‘tuberculosis’. A paper published in 1934 titled ‘The Association of Diabetes and Tuberculosis’ notes that “in 1883 Bouchardat, the great French student of diabetes, stated in his text that at autopsy every case of diabetes had tubercles in the lungs. Earlier in the century Griesinger had found tuberculosis in 42 per cent of 250 diabetics, and Frerichs found in 55 autopsies 21 cases of pulmonary tuberculosis. Windle is frequently quoted as stating that in over 50 per cent of 333 diabetic autopsies pulmonary tuberculosis was found.” There also appears to be an association with ‘syphilis’; “diabetes not uncommonly follows syphilitic infection”, and even ‘chickenpox’; “the paper describes two individuals presenting with acute insulin dependent diabetes mellitus for a brief and transient period. Both had had chicken pox infection in the immediate past.” We have previously discussed these ‘Victorian diseases’ in more detail here, and like ‘scurvy’, have linked them to poisoning.
In 1922, Leonard Thompson, a 14-year old boy with Type 1 diabetes, became the first person to receive an injection of insulin. Before then, according to Harvard Medical, “type 1 diabetes was a death sentence: half of people who developed it died within two years; more than 90% were dead within five years”.
Prior to this, ‘starvation diets’ where advocated for by American physicians Frederick Allen and Elliott Joslin, “to prolong the lives of diabetic patients”.
Given what we have read with regards to Dr. Mott’s treatment of his patient, one does wonder however whether the death was brought about as a result of lack of insulin, or as a result of being prescribed various poisons, such as arsenic. Indeed, it would appear Dr. Mott was not the only one to attempt such a treatment; “from the late 1700s, arsenic was first tested as a cure for a variety of diseases and symptoms … In 1873, La France Medicale published that several doctors treated their diabetic patients with arsenic resulting in diminished sugar in the urine … Several other doctors who did not have success with the treatment method were described of having patients with different types of diabetes”.
And so without further ado, we turn our attention to the King of Poisons.
Arsenic
We have already discussed in detail how arsenic disrupts a number of bodily processes, resulting in a wide range of seemingly disparate ‘diseases’ said to be caused by various microorganisms, autoimmune disorders, genetics and so forth. A literature review published in 2017 states that “arsenic affects almost all vital organs of human body causing the damage or dysfunction”, and a literature review published in 2020 states that “a large body of evidence has shown a link between arsenic exposure and diabetes, but the underlying mechanisms have not yet been clarified”.
As previously discussed in ‘From the gut with love’, arsenic has an affinity for sulphur, and as discussed here, both insulin and cell receptors are sulphur-rich. According to a paper published in 2004 by Tseng “arsenite has high affinity for sulfhydryl groups and thus can form covalent bonds with the disulfide bridges in the molecules of insulin, insulin receptors, glucose transporters (GLUTs), and enzymes involved in glucose metabolism. As a result, the normal functions of these molecules can be hampered”.
It is worth reminding ourselves that it is well established that arsenic has been found to bioaccumulate in all the major organs, in a dose-dependent way, including the heart. It would appear other organs are not exempt. According to a paper published in 2007, mice exposed to arsenic in their diet were found to have “increases in the concentration of iAs and its metabolites in the liver and in organs targeted by type 2 diabetes, including pancreas, skeletal muscle and adipose tissue”. A literature review published in 2021 re-affirms this; “arsenic is taken up by pancreatic tissue and β-cells in a dose-dependent manner, and accumulated significantly in the pancreas”.
The literature cited thus far appears to be rather confused as to what the cause(s) of diabetes are. We will now examine in turn, the key markers associated with diabetes, and whether arsenic exposure may be able to explain them.
Arsenic, obesity & pathogenic microorganisms
Type 2 diabetes is often said be the result of lifestyle choices that result in obesity. According to a literature review published in 2022, there is evidence of increased BMI in individuals exposed to arsenic, although other studies have found no association. It is interesting to note however, that arsenic-based compound Roxarsone was approved in 1944 by the FDA as a chicken-feed additive to promote growth, and more importantly, give the meat an “appealing pink colour”. As VOX notes “Chickens have gotten ridiculously large since the 1950s”. Yes indeed.
According to paper published in 1956, the growth promoting potential of arsenic-based compounds was first recognised in 1949. An article published by the Union of Concerned Scientists states that “by 2000, an estimated 66 percent of broiler chicken producers used roxarsone additives in feed to enhance growth and control coccidiosis, a parasitic disease found in chickens”. In 2011, the FDA published a report where they reported that “Roxarsone and its metabolites were present in liver tissues from chicks fed roxarsone-medicated feed”. The findings were confirmed by later studies, such as Nachman et al. 2013, where they found that “the use of arsenic-based drugs in chicken production results in dietary exposures to iAs”.
Certain species of bacteria have been found to “transform the roxarsone in the litter releasing the more toxic inorganic arsenic”. The researchers at the time noted that this can lead to “can lead to increased soil arsenic concentration and arsenic contaminated dust … Clostridium and Lactobacillus species are common in the chicken cecum and litter”. In 2017, McGill published an article on the subject where the author asks the question; “what happens to the vast amount of poop that is produced by the over ten billion chickens slaughtered every year”? According to them; “most of it gets spread on crop land as fertilizer or is formulated into pellets for use by the home gardener”.
This suggests that arsenic does indeed have an obesogenic effect, which in turn may explain why certain ‘processed foods’ are said to have this property. This may help Hurley resolve the ‘mystery’ of why historical accounts of diabetes appear to make no mention of the weight of the individual; obesity is a symptom, not a cause, and apparent correlation does not automatically imply causation. A paper published in 2016 notes that “the proportion of people with type 2 diabetes who are of normal BMI has decreased as the prevalence of overweight/obesity has increased, caused by the exponential relationship between BMI and the prevalence of type 2 diabetes”.
We have previously discussed at length the problems with the study of ‘viruses’, bacteria, and other microorganisms, and the claims that they are ‘pathogenic’. ‘Viruses’, as discussed in ‘A hitchikers guide to exosomes’ are indistinguishable from so-called ‘exosomes’. In 2022, researchers found more ‘exosomes’ in fish exposed to various ‘environmental pollutants’, including arsenic and mercury, and noted that “the increased circulating exosome level of indigenous fish samples was consistent with the oxidative damage and genotoxic effects of the water environment”. The study concludes that “circulating exosome levels may be novel biomarker for ecotoxic effects of water environment”, which in turn raises the question of whether so-called ‘viral load’ is in reality, an indicator of ‘toxic load’.
As a reminder, diet / lifestyle, and ‘viral triggers’ are speculated to be the causes of the two ‘types’ of diabetes. Both can be explained by arsenic exposure. So, let’s now move on to see how arsenic affects the relevant parts of the body.
Arsenic, insulin cell receptors, and insulin
In its fact sheet on arsenic, the CDC references a study by Rossman published in 2007 where “it is hypothesized that arsenic’s diabetogenic effect may be related to its ability to bind and inhibit the insulin receptor”. We were unable to locate this paper, however others appear to confirm this. In a literature review that published in 2021 by Javaid et al., the authors write that “arsenic binds and renders nonfunctional insulin and insulin receptor bridge (disulfide)”.
Experimental evidence appears to confirm this. In 2012, a research team created ‘nano-insulin’ – insulin that was ‘nano-encapsulated’ with PLGA (Poly(lactic-co-glycolic) acid). The team found that this ‘nano-insulin’ “had less distorted secondary structure as compared with that of insulin in presence of arsenic”. They concluded that “Nano insulin could hinder the high affinity of arsenite for exofacial sulphydryl groups and the covalent bonds formed by the interaction of arsenite with the disulfide bridges in the molecules of insulin receptors, glucose transporters (GLUTs) and enzymes involved in glucose metabolism more efficiently than insulin alone”.
Arsenic & ATP inhibition
In the aforementioned paper, Tseng also noted that arsenate can displace phosphate in the formation of ATP, as well as the phosphates involved in glucose metabolism “which could theoretically slow down the normal metabolism of glucose, interrupt the production of energy, and interfere with the ATP-dependent insulin secretion”. Although he notes that “the concentration of arsenate required for such reaction is high and not physiologically relevant”, some studies appear to have demonstrated that this can indeed happen; a literature review published in 2014 notes that “an in vitro study suggested that arsenate exposure caused a reduction in ATP in rabbit and human erythrocytes [red blood cells]”.
The aforementioned literature review also notes that “arsenic acts as a competent of phosphate ion (Pi) and interacts with adenosine-diphosphate to produce adenosine-diphosphate arsenate (ADPA) that is then applied to production of glucose-6-arsenate”.
Arsenic & T cells
A study published in 2020 confirmed the results of previous studies that had found how arsenic affects Treg cell levels. Tregs cells are a type of T cell that exist to regulate immune responses; “it has been shown that Tregs are able to inhibit T cell proliferation and cytokine production and play a critical role in preventing autoimmunity”.
The researchers found that “with increasing NaAsO2 concentrations, the proportion of Tregs was obviously elevated in comparison to the control group”.
Arsenic & the liver
Like the pancreas, numerous studies have found that arsenic and it’s metabolites accumulate in the liver (and this includes the livers of chickens fed with arsenic-based compounds). All endogenous insulin flows through the liver. If insulin is damaged by arsenic, then it seems possible that the liver may be one location where the damage is occurring, which in turn might explain why exogenous insulin is not affected in the same way; as previously discussed it doesn’t flow through the liver.
Arsenic & zinc fingers
A study published in 2006 found that ‘zinc finger’ proteins are “essential for the development of pancreatic beta cells and intestinal endocrine cells”. We have previously discussed in ‘Sonic Hedgehog and the Iron Fortress’ the importance of zinc in controlling what flows in and out of cells, and how zinc can be displaced by arsenic. Experimental evidence appears to confirm this. In 2019, a study was published that found that “zinc deficiency and arsenic, both independently and in combination, adversely affect pancreatic beta cell health”.
Arsenic, beta cell death, and dysfunction
Arsenic appears to affect both beta cell mass, and function in a number of different ways. In a study published in 2011, researchers found that arsenic can “reduce purinergic Ca2+ signaling in a dose-dependent manner”. If you recall, the calcium channels control how much insulin is released from the beta cells.
Some studies have also found that arsenic can induce beta cell death. For instance, a study published in 2018 found that “iAS [inorganic arsenic] and atorvastatin, alone or together, are capable of activating an apoptotic process in pancreatic β-cells via caspase-3”. Others have found that “pancreatic β-cell mass was not altered, suggesting that arsenic primarily disrupts β-cell function”.
A literature review published in 2011 also notes that “pancreatic β-cells are the most vulnerable to oxidative stress-induced damage because they have lower levels of antioxidant enzymes”. The aforementioned review by Javaid et al. also notes that “this weak defense system makes β cells extremely sensitive to heavy metals and their effects, resulting in the dysfunction, destruction, or even death of pancreatic β cells”.
A study published in 2010 found that prolonged exposure of beta cells to low levels of arsenite led to “decreased GSIS [glucose-stimulated insulin secretion] in a dose-dependent manner”. According to the researchers, this was caused by the action of ‘reactive oxygen species’; “GSH and H2O2-scavenging activity were significantly increased in the arsenite-treated cells … The increased antioxidant activity significantly inhibited net glucose-stimulated intracellular peroxide production, which is involved in GSIS … These studies suggest that low levels of arsenic trigger a cellular adaptive response, which impairs ROS signaling involved in GSIS, and thus disturbs β-cell function”.
Researchers in 2020 also found that arsenic caused “execution of ferroptosis in a dose-dependent manner”, which in turn induced “β cells dysfunction”. Ferroptosis, according to a literature review published in 2021 “is a type of regulated necrosis that is triggered by a combination of iron toxicity, lipid peroxidation, and plasma membrane damage”. According to the authors, “excessive iron accumulation” is one of the triggers for ferroptosis. This is discussed in more detail in The Iron Key.
Discussion
A paper published in 2011 suggested that diabetes does not refer to fundamentally different ‘diseases’, but rather a “continuous spectrum”. The authors note this is required because “of the accumulating evidence of immune system involvement in the pathophysiology of type 2 diabetes”.
It seems like the authors of this paper are on to something. If, as we suspect, arsenic is the primary source of the problem, then indeed, diabetes can be thought of as a spectrum – a spectrum of damage. Although arsenic appears to be a prime suspect, it is also interesting to note that ionising radiation also appears to be a trigger. In a study published in 2004, researchers found “a significant and documented increase in the incidence of Type 1 diabetes in children and adolescents after Chernobyl in the radioactively contaminated area of Gomel compared to Minsk”. This appears to have been confirmed by studies on rats; irradiation resulted in a “reduced number of beta-cells by islet”. We have previously linked both arsenic and ionising radiation to COVID, which may in turn explain the observations made by clinicians throughout the ‘pandemic’.
As mentioned previously arsenic has an affinity for sulphur, meaning it will readily bind with any sulphur that is contained, for instance, in wheat. This in turn may explain why the clinicians in Denmark appear to have had some success treating their patient with a gluten-free diet. It is also interesting to note that sulfonylureas are also used as herbicides. A study published in 2009 noted that these compounds have the ability to bind to heavy metals. Given that these tablets are typically taken at meal times, is the purpose of them to provide a binding target for any ingested arsenic in order to give endogenous insulin a chance to do what it needs to do?
Diabetes is often linked to a number of other diseases said to be ‘autoimmune’, including for instance, psoriasis, rheumatoid arthritis, and thyroid problems. We have linked arsenic exposure to all of these, and a more detailed article on how arsenic impacts the thyroid is available here. Another form of diabetes we haven’t discussed in detail is ‘Cystic fibrosis-related diabetes’, which is said to have “features of both type 1 and type 2”, and is therefore considered a different condition. Researchers in 2015 linked the development of cystic fibrosis to a “genetic defect in the CFTR protein which regulates the flow of chloride in and out of cells”. Coincidentally, “recent studies in cell culture show that arsenic causes the CFTR protein to break down”.
We haven’t discussed ‘gestational diabetes’ in detail – diabetes that appears during pregnancy. A study published in 2015, concluded that “exposure to some of the selected metals (noticeably As [arsenic]) may contribute to maternal GDM risk during pregnancy”.
Although this article does not provide any quick fixes, it is written with the hope that by honing in on what appears to be the fundamental problem – not just for diabetes, but a number of other ‘diseases’, efforts can be directed at developing long-term solutions. Some will say that the only solution will eventually come from stem cell therapy. As previously mentioned, it is interesting to note that contratory to popular belief, beta cells appear to be continuously regenerated. In a paper published in 2005, the authors conclude that “the presence of beta cells despite ongoing apoptosis implies, by definition, that concomitant new beta cell formation must be occurring, even after long-standing type 1 diabetes. We conclude that type 1 diabetes may be reversed by targeted inhibition of beta cell destruction”.
Another paper published in 2019 notes that “an important insight which is now gaining widespread acceptance is that, even in patients with long-standing T1D, there is a functional reserve of β-cells. This is a surprising finding which has challenged the long-held dogma that T1D only develops when 80–90% of β-cells are lost…Moreover, analysis of C-peptide levels in patients living with T1D for increasing periods has provoked the surprising conclusion that the rate of β-cell loss is initially exponential but then declines dramatically at about 7 years post-diagnosis such that β-cell mass remains stable thereafter”. In the aforementioned animal study, researchers found that in rats from which 90% of the pancreas was removed, “there is substantial β-cell regeneration in these rats such that β-cell mass 10–12 weeks after the surgery is about 40% of normal”.
This in turn shows that the body has an incredible ability to heal itself, if given the opportunity to. As noted previously, before the advent of insulin, fasting was advocated for as a ‘cure’. Could this have helped in some instances? Fasting has been found to provide a good way of clearing arsenic from the body. A study published in 2007 concluded that “fasting for a period of 12 h results in a significant increase in the percentage of urinary arsenic as MA, and its frequency of detection in the volunteers at the end of the fasting period is almost nine fold higher”. Another study published in 2021 found that “fasting caused a decrease in body weight, and in urinary arsenic (by 72%) and nickel (by 15%) concentrations. A decrease in lead hair concentrations (by 30%) was documented”. Some studies appear to have already shown some promising results. A literature review published in 2021 notes that “current evidence suggests that intermittent fasting is an effective non-medicinal treatment option for type 2 diabetes”. Less research is available for Type 1, because it would appear there is a widely held belief that this should not be attempted; “it is universally agreed that people with type 1 diabetes should not attempt intermittent fasting”. However, researchers in 2021 found that it is possible for Type 1 diabetics to fast safely; “our study showed that people with type 1 diabetes can safely perform a 36 hours fasting period with a low risk of hypoglycemia and ketoacidosis” (please note that anyone attempting this should first seek counsel from their doctor).
As this article has hopefully demonstrated, there is a significant amount of evidence and body of literature that not only links arsenic to the development of diabetes, but can explain at a molecular level, how. In spite of this, health authorities such as the WHO continue to say that the causes of diabetes are ‘unknown’. It is also interesting to note that according to a paper published in June 2022, a considerable body of literature has been developing in the last 30 or so years focusing on how to ‘remedy’ the global arsenic situation.
According to a literature review published in 2020, many of these papers have focused on the impact arsenic has on metabolism, and increasingly DNA damage and cancer.
Thank you as always to Caroline for her invaluable help in piecing this information together.